Morrison, B. E., Park, S. J., Mooney, J. M. & Mehrad, B. Chemokine-mediated recruitment of NK cells is a critical host defense mechanism in invasive aspergillosis. J. Clin. Investig. 112, 1862–1870 (2003).
Marolda, A. et al. Candida species-dependent release of IL-12 by dendritic cells induces different levels of NK cell stimulation. J. Infect. Dis. https://doi.org/10.1093/infdis/jiaa035 (2020).
Stuehler, C. et al. Immune reconstitution after allogeneic hematopoietic stem cell transplantation and association with occurrence and outcome of invasive aspergillosis. J. Infect. Dis. 212, 959–967 (2015).
Schmidt, S. et al. Human natural killer cells exhibit direct activity against Aspergillus fumigatus hyphae, but not against resting conidia. J. Infect. Dis. 203, 430–435 (2011).
Park, S. J., Hughes, M. A., Burdick, M., Strieter, R. M. & Mehrad, B. Early NK cell-derived IFN-γ is essential to host defense in neutropenic invasive aspergillosis. J. Immunol. 182, 4306–4312 (2009).
Bouzani, M. et al. Human NK cells display important antifungal activity against Aspergillus fumigatus, which is directly mediated by IFN-release. J. Immunol. 187, 1369–1376 (2011).
Li, S. S. et al. The NK receptor NKp30 mediates direct fungal recognition and killing and is diminished in NK cells from HIV-infected patients. Cell Host Microbe 14, 387–397 (2013).
Vitenshtein, A. et al. NK cell recognition of Candida glabrata through binding of NKp46 and NCR1 to fungal ligands Epa1, Epa6, and Epa7. Cell Host Microbe 20, 527–534 (2016).
Charpak-Amikam, Y. et al. The activating receptor NKG2D is an anti-fungal pattern recognition receptor. Nat. Commun. 15, 8664 (2024).
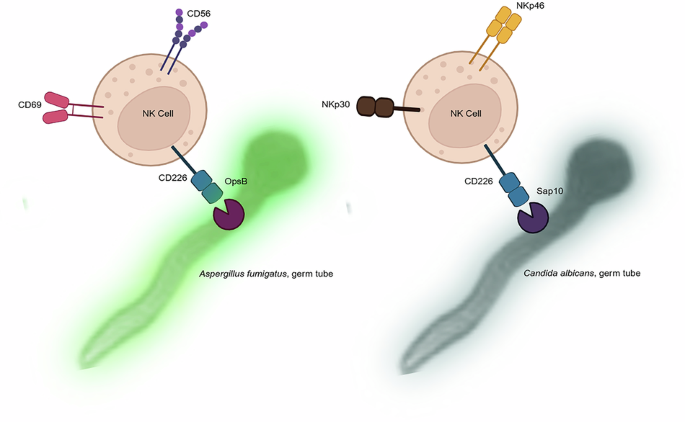
Heilig, L. et al. CD56-mediated activation of human natural killer cells is triggered by Aspergillus fumigatus galactosaminogalactan. PLOS Pathog 20, e1012315 (2024).
Ziegler, S. et al. CD56 is a pathogen recognition receptor on human natural killer cells. Sci. Rep. 7, 6138 (2017).
Enqvist, M. et al. Coordinated expression of DNAM-1 and LFA-1 in educated NK cells. J. Immunol. 194, 4518–4527 (2015).
Martinet, L. & Smyth, M. J. Balancing natural killer cell activation through paired receptors. Nat. Rev. Immunol. 15, 243–254 (2015).
Huang, Z., Qi, G., Miller, J. S. & Zheng, S. G. CD226: An emerging role in immunologic diseases. Front. Cell Dev. Biol. 8, 564 (2020).
Martinet, L. et al. DNAM-1 expression marks an alternative program of NK cell maturation. Cell Rep 11, 85–97 (2015).
Yeo, J., Ko, M., Lee, D.-H., Park, Y. & Jin, H. TIGIT/CD226 axis regulates anti-tumor immunity. Pharmaceuticals 14, 200 (2021).
Tahara-Hanaoka, S. et al. Identification and characterization of murine DNAM-1 (CD226) and its poliovirus receptor family ligands. Biochem. Biophys. Res. Commun. 329, 996–1000 (2005).
Chan, C. J. et al. The receptors CD96 and CD226 oppose each other in the regulation of natural killer cell functions. Nat. Immunol. 15, 431–438 (2014).
Albrecht, A. et al. Glycosylphosphatidylinositol-anchored proteases of Candida albicans target proteins necessary for both cellular processes and host-pathogen interactions. J. Biol. Chem. 281, 688–694 (2006).
Schild, L. et al. Proteolytic cleavage of covalently linked cell wall proteins by Candida albicans Sap9 and Sap10. Eukaryot. Cell 10, 98–109 (2011).
Hou, S., Zheng, X., Wei, H., Tian, Z. & Sun, R. Recombinant soluble CD226 protein directly inhibits cancer cell proliferation in vitro. Int. Immunopharmacol. 19, 119–126 (2014).
Kim et al. CD226-/- natural killer cells fail to establish stable contacts with cancer cells and show impaired control of tumor metastasis in vivo. Oncoimmunology 5, e1338994 (2017).
Chiang, E. Y. & Mellman, I. TIGIT-CD226-PVR axis: advancing immune checkpoint blockade for cancer immunotherapy. J. Immunother. Cancer 10, e004711 (2022).
Stannard, K. A. et al. Human peripheral blood DNAM-1neg NK cells are a terminally differentiated subset with limited effector functions. Blood Adv 3, 1681–1694 (2019).
Weiss, E. et al. Reconstituting NK cells after allogeneic stem cell transplantation show impaired response to the fungal pathogen Aspergillus fumigatus. Front. Immunol. 11, 2117 (2020).
Murillo, O. et al. Costimulatory CD226 signaling regulates proliferation of memory-like NK cells in healthy individuals with latent mycobacterium tuberculosis infection. Int. J. Mol. Sci. 23, 12838 (2022).
Jafari, M., Mirzaie, M. & Sadeghi, M. Interlog protein network: an evolutionary benchmark of protein interaction networks for the evaluation of clustering algorithms. BMC Bioinformatics 16, 319 (2015).
Mosca, R., Céol, A., Stein, A., Olivella, R. & Aloy, P. 3did: a catalog of domain-based interactions of known three-dimensional structure. Nucleic Acids Res. 42, D374–D379 (2014).
Shimizu, N. et al. Oryzapsins, the orthologs of yeast yapsin in Aspergillus oryzae, affect ergosterol synthesis. Appl. Microbiol. Biotechnol. 105, 8481–8494 (2021).
Abramson, J. et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 630, 493–500 (2024).
Pierce, B. G. et al. ZDOCK server: interactive docking prediction of protein–protein complexes and symmetric multimers. Bioinformatics 30, 1771–1773 (2014).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Krissinel, E. & Henrick, K. Inference of macromolecular assemblies from crystalline state. J. Mol. Biol. 372, 774–797 (2007).
de Castro, E. et al. ScanProsite: detection of PROSITE signature matches and ProRule-associated functional and structural residues in proteins. Nucleic Acids Res. 34, W362–W365 (2006).
Steentoft, C. et al. Precision mapping of the human O-GalNAc glycoproteome through SimpleCell technology. EMBO J. 32, 1478–1488 (2013).
Chauhan, J. S., Rao, A. & Raghava, G. P. S. In silico platform for prediction of N-, O- and C-glycosites in eukaryotic protein sequences. PLoS ONE 8, e67008 (2013).
Jo, S., Kim, T., Iyer, V. G. & Im, W. CHARMM-GUI: a web-based graphical user interface for CHARMM. J. Comput. Chem. 29, 1859–1865 (2008).
Deuss, F. A. et al. Structural basis for the recognition of nectin-like protein-5 by the human-activating immune receptor, DNAM-1. J. Biol. Chem. 294, 12534–12546 (2019).
Ge, Z., Peppelenbosch, M. P., Sprengers, D. & Kwekkeboom, J. TIGIT, the next step towards successful combination immune checkpoint therapy in cancer. Front. Immunol. 12, (2021).
Feng, S., Isayev, O., Werner, J. & Bazhin, A. V. CD96 as a potential immune regulator in cancers. Int. J. Mol. Sci. 24, 1303 (2023).
Shibuya, A. et al. DNAM-1, a novel adhesion molecule involved in the cytolytic function of T lymphocytes. Immunity 4, 573–581 (1996).
Cibrián, D. & Sánchez-Madrid, F. CD69: from activation marker to metabolic gatekeeper. Eur. J. Immunol. 47, 946–953 (2017).
Mandala, W., Harawa, V., Munyenyembe, A., Soko, M. & Longwe, H. Optimization of stimulation and staining conditions for intracellular cytokine staining (ICS) for determination of cytokine-producing T cells and monocytes. Curr. Res. Immunol. 2, 184–193 (2021).
Rodrigo, M. B. et al. Dual fluorescence reporter mice for Ccl3 transcription, translation, and intercellular communication. J. Exp. Med. 221, e20231814 (2024).
Liu, S. et al. NK cell-based cancer immunotherapy: from basic biology to clinical development. J. Hematol. Oncol. 14, 7 (2021).
Zhang, Z. et al. DNAM-1 controls NK cell activation via an ITT-like motif. J. Exp. Med. 212, 2165–2182 (2015).
Mattiola, I. Immune circuits to shape natural killer cells in cancer. Cancers 13, 3225 (2021).
Shirakawa, J. et al. LFA-1-dependent lipid raft recruitment of DNAM-1 (CD226) in CD4+ T cell. Int. Immunol. 18, 951–957 (2006).
Shirakawa, J., Shibuya, K. & Shibuya, A. Requirement of the serine at residue 329 for lipid raft recruitment of DNAM-1 (CD226). Int. Immunol. 17, 217–223 (2005).
Cifaldi, L. et al. DNAM-1 activating receptor and its ligands: how do viruses affect the NK cell-mediated immune surveillance during the various phases of infection? Int. J. Mol. Sci. 20, 3715 (2019).
Valentine, M. et al. Nanobody-mediated neutralization of candidalysin prevents epithelial damage and inflammatory responses that drive vulvovaginal candidiasis pathogenesis. mBio 15, e03409-23 (2024).
Dutton, L. C., Jenkinson, H. F., Lamont, R. J. & Nobbs, A. H. Role of Candida albicans secreted aspartyl protease Sap9 in interkingdom biofilm formation. Pathog. Dis. 74, ftw005 (2016).
Kadry, A. A., El-Ganiny, A. M. & El-Baz, A. M. Relationship between Sap prevalence and biofilm formation among resistant clinical isolates of Candida albicans. Afr. Health Sci. 18, 1166–1174 (2018).
Naglik, J. R., Challacombe, S. J. & Hube, B. Candida albicans secreted aspartyl proteinases in virulence and pathogenesis. Microbiol. Mol. Biol. Rev. MMBR 67, 400–428 (2003).
Ibe, C. & Munro, C. A. Fungal cell wall proteins and signaling pathways form a cytoprotective network to combat stresses. J. Fungi 7, 739 (2021).
Irmer, H. et al. RNAseq analysis of Aspergillus fumigatus in blood reveals a just wait and see resting stage behavior. BMC Genom. 16, 640 (2015).
van Kooyk, Y. & Rabinovich, G. A. Protein-glycan interactions in the control of innate and adaptive immune responses. Nat. Immunol. 9, 593–601 (2008).
Lin, B., Qing, X., Liao, J. & Zhuo, K. Role of protein glycosylation in host-pathogen interaction. Cells 9, 1022 (2020).
Tahara, S., Okumura, G., Matsuo, T., Shibuya, A. & Shibuya, K. Essential role of CD155 glycosylation in functional binding to DNAM-1 on natural killer cells. Int. Immunol. 36, 317–325 (2024).
Gaud, G. et al. The costimulatory molecule CD226 signals through VAV1 to amplify TCR signals and promote IL-17 production by CD4+ T cells. Sci. Signal. 11, eaar3083 (2018).
Dastouri, M., Kilic, N. & Yilmaz, H. The apoptotic effects of NK-92 cells stimulated with an anti-CD226 antibody on MDA-MB-231 triple-negative breast cancer cells. Med. Oncol. 40, 228 (2023).
Schmidt, S., Zimmermann, S.-Y., Tramsen, L., Koehl, U. & Lehrnbecher, T. Natural killer cells and antifungal host response. Clin. Vaccine Immunol. 20, 452–458 (2013).
Bernardini, G. et al. CCL3 and CXCL12 regulate trafficking of mouse bone marrow NK cell subsets. Blood 111, 3626–3634 (2008).
Seymour, F., Cavenagh, J. D., Mathews, J. & Gribben, J. G. NK cells CD56bright and CD56dim subset cytokine loss and exhaustion is associated with impaired survival in myeloma. Blood Adv. 6, 5152–5159 (2022).
Montazersaheb, S., Fathi, E. & Farahzadi, R. Cytokines and signaling pathways involved in differentiation potential of hematopoietic stem cells towards natural killer cells. Tissue Cell 70, 101501 (2021).
Sharma, R. & Das, A. IL-2 mediates NK cell proliferation but not hyperactivity. Immunol. Res. 66, 151–157 (2018).
Deng, Y. et al. Transcription factor Foxo1 is a negative regulator of natural killer cell maturation and function. Immunity 42, 457–470 (2015).
Gilfillan, S. et al. DNAM-1 promotes activation of cytotoxic lymphocytes by nonprofessional antigen-presenting cells and tumors. J. Exp. Med. 205, 2965–2973 (2008).
Marangio, C. et al. NKG2D triggering hampers DNAM-1-mediated signaling in human NK cells. Front. Immunol. 16, 1575059 (2025).
Charpak-Amikam, Y. et al. Candida albicans evades NK cell elimination via binding of agglutinin-like sequence proteins to the checkpoint receptor TIGIT. Nat. Commun. 13, 2463 (2022).
Margraf-Schönfeld, S., Böhm, C. & Watzl, C. Glycosylation affects ligand binding and function of the activating natural killer cell receptor 2B4 (CD244) protein. J. Biol. Chem. 286, 24142–24149 (2011).
Lin, Y.-X., Hung, M.-C., Hsu, J.-L. & Hsu, J.-M. The N-linked glycosylations of TIGIT Asn32 and Asn101 facilitate PVR/TIGIT interaction. Biochem. Biophys. Res. Commun. 562, 9–14 (2021).
Qin, Y. et al. Upregulation of CD226 on subsets of T cells and NK cells is associated with upregulated adhesion molecules and cytotoxic factors in patients with tuberculosis. Int. Immunopharmacol. 120, 110360 (2023).
Ding, Y. et al. Distinct developmental pathways generate functionally distinct populations of natural killer cells. Nat. Immunol. 25, 1183–1192 (2024).
Trinks, N. et al. Subdiffraction-resolution fluorescence imaging of immunological synapse formation between NK cells and A. fumigatus by expansion microscopy. Commun. Biol. 4, 1151 (2021).
Ruf, D., Brantl, V. & Wagener, J. Mitochondrial fragmentation in Aspergillus fumigatus as early marker of granulocyte killing activity. Front. Cell. Infect. Microbiol. 8, 128 (2018).
Hess, S. T., Huang, S., Heikal, A. A. & Webb, W. W. Biological and chemical applications of fluorescence correlation spectroscopy: a review. Biochemistry 41, 697–705 (2002).
Schüttpelz, M. et al. Changes in conformational cynamics of mRNA upon At GRP7 binding studied by fluorescence correlation spectroscopy. J. Am. Chem. Soc. 130, 9507–9513 (2008).
The UniProt Consortium. UniProt: the universal protein knowledgebase in 2021. Nucleic Acids Res. 49, D480–D489 (2021).
The Gene Ontology Consortium Gene Ontology Consortium: going forward. Nucleic Acids Res. 43, D1049–D1056 (2015).












Leave a Reply