Venetoclax directly enhances the effector function of murine CD19CART cells
Based upon our previous work showing that T cells adapt to BCL-2 drugging through adaptive effector-like changes, we sought to determine if venetoclax could amplify the efficacy of adoptively expanded T cells using CART as a model system. To first test this, we used a well-described murine T cell model engineered to express a murine anti-CD19 CAR (mCD19CART).30,31 Here, CD45.1 transgenic mice received vehicle or venetoclax 50 mg/kg for 15 days (5 days/week for 3 weeks) in an effort to provide long-term BCL-2 drugging as tested in our previous murine studies.24,25 T cells from these animals were isolated, activated, and transduced to express anti-murine CD19CAR and expanded ex vivo in the presence or absence of venetoclax (Supplementary Fig. S1a). We then examined the effector function of these mCD19CART through an in vitro cytotoxicity assay where differently treated mCD19CART were co-cultured with CD19+ murine B cell lymphoma (A20) at various effector to tumor (E:T) ratios. We found that administration of venetoclax either in vivo, ex vivo, or both resulted in enhanced antitumor mCD19CART killing of target cells compared to vehicle-treated CART (Supplementary Fig. S1b). However, ex vivo treatment with venetoclax of in vivo vehicle-treated cells during manufacturing resulted in the greatest enhancement of mCD19CART effector functionality (Supplementary Fig. S1c). Importantly, venetoclax did not affect CAR expression or the memory phenotype of the final CART product (Supplementary Fig. S1d, e). These results suggest that expansion of CART under BCL-2 blockade with venetoclax can increase CART-mediated tumor killing in a manner not related to apoptosis.
BCL-2 blockade is compatible with human CART cell manufacturing
We next aimed to translate the previous findings to human CART cells. Given that BH3 mimetics were originally designed to induce target cell apoptosis,32 we wondered whether venetoclax treatment would negatively interfere with human CD19CART (hereafter referred to as CART) production by inducing cell death and limiting CART cell proliferation and expansion. To test this, following activation and CD19CAR lentivirus transduction, healthy donor CART were expanded with escalating doses of venetoclax (Fig. 1a). The dosing range chosen for treatment is equivalent to the plasma drug concentration measured in patients following standard dosing.33 Venetoclax resulted in a dose-dependent but non-statistically significant decrease in the number of cells in the final CART product (Fig. 1b). Taken together, this data suggests that venetoclax, by virtue of specific targeting of BCL-2, is a suitable BH3 mimetic to be tested and combined with CART manufacturing.
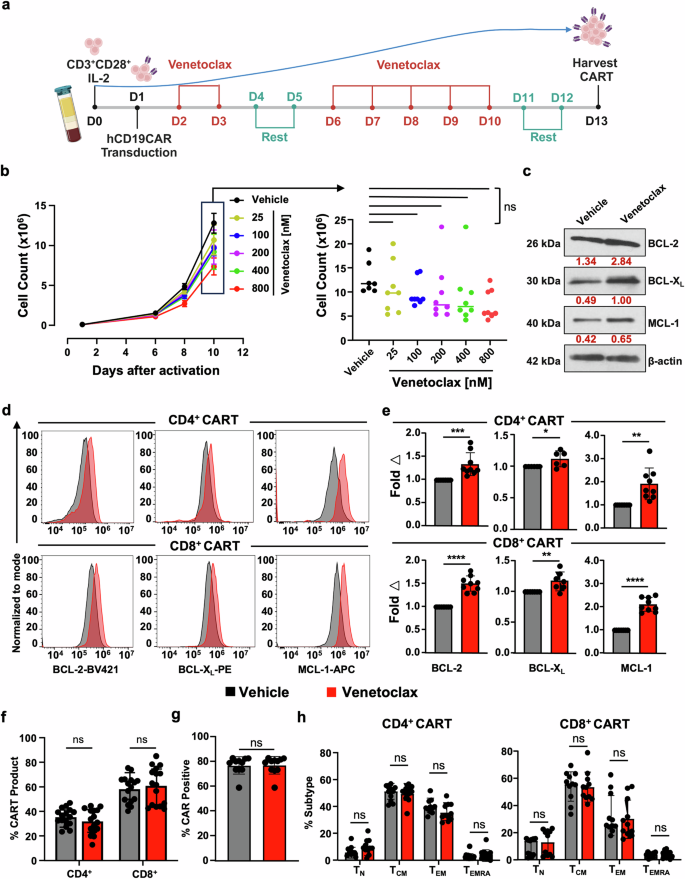
Venetoclax treatment is tolerated during CART expansion and induces upregulation of anti-apoptotic protein expression. a Schema of CD19CART (CART) production. T cells were isolated from PBMCs from healthy donors on day 0 and activated overnight with human anti-CD3/CD28 beads, followed by transduction with lentivirus designed to express hCD19CAR on day 1. On day 2, cells were either treated with vehicle (DMSO) or with the indicated doses of venetoclax daily until day 13. b CART treated with increasing doses of venetoclax shows no significant changes in T cell expansion compared to vehicle-treated CART. On left: proliferation curve indicating CART cell count for each treatment condition at different time points. On the right: comparison of CART cell counts on day 10 following treatment with increasing doses of venetoclax. c–e CART cells have increased anti-apoptotic protein expression upon expansion in the presence of venetoclax on day 13 of production. c BCL-2 family anti-apoptotic protein levels were evaluated in total vehicle vs venetoclax-treated CART by Western blot. Expression values (red) were normalized to the corresponding β-actin. d Protein expression measured using intracellular flow cytometric evaluation of BCL-2, BCL-XL, and MCL-1 in venetoclax-treated CART (red) and vehicle-treated CART (black). e BCL-2 family protein expression in venetoclax-treated CART normalized to expression in vehicle-treated CART. Venetoclax-treated CARTs show no significant differences in CART (f) CD4:CD8 ratio, g CAR+ expression, or h T cell memory phenotype. TN: naïve (CCR7+CD45RA+), TCM: central memory (CCR7+CD45RA−), TEM: effector memory (CCR7−CD45RA−), and TEMRA: terminally differentiated (CCR7−CD45RA+). Data are from at least three independent experiments from different healthy donors (n = 3) with two-three technical replicates in bar graphs and shown as mean ± SD. One-way ANOVA followed by Tukey’s multiple comparison tests was used for (b), unpaired Student’s t-test for (e–g), and two-way ANOVA followed by SIDAK’s multiple comparison tests for (h). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns non-significant. Image (a) Created in BioRender
Venetoclax-treated CART upregulates anti-apoptotic BCL-2 protein levels
We previously found that in vitro and in vivo treatment of murine CD4+, CD8+, and FOXP3+ regulatory T cells with venetoclax resulted in increased protein levels of BCL-2, BCL-XL, and MCL-1.24 Aligned with this phenomenon, CART expanded in the presence of venetoclax also showed upregulation of BCL-2, BCL-XL, and MCL-1 proteins in total CART (Fig. 1c) and similarly in CD4+ and CD8+ CART subsets (Fig. 1d, e). Like our initial studies in mCD19CART (Supplementary Fig. S1), venetoclax did not affect the CD4+/CD8+ ratio, CAR expression, or the memory population composition of the final CART product when compared to vehicle-treated cells (Fig. 1f–h). Collectively, these data indicate that ex vivo pressure on the apoptotic machinery via specific BCL-2 blockade results in compensatory upregulation of anti-apoptotic proteins without significantly affecting CART composition or expansion.
Venetoclax enhances CART cytotoxicity in vitro
Recent reports indicate that treating conventional or double-negative patient-derived T cells with venetoclax enhances their anti-leukemic activity through poorly understood mechanisms.22 Since our venetoclax-treated mCD19CART showed improved antitumor efficacy following ex vivo treatment (Supplementary Fig. S1), we wondered if venetoclax-treated human CART would have similarly enhanced activity even with their longer ex vivo expansion. To test this, we performed killing assays using vehicle or venetoclax-treated CART cocultured with the CD19+ expressing human diffuse large B cell lymphoma (DLBCL) cell line OCI-Ly8. Aligned with our previous findings, venetoclax-treated CART dose-dependently killed tumor cells more effectively than vehicle-treated CART (Fig. 2a). Importantly, venetoclax was not present in the cultures, thus ensuring that target cell death was not the result of direct BCL-2 drugging within tumor cells. Similarly, increased killing was observed against the CD19+ acute lymphoblastic leukemia cell line NALM6 (Fig. 2b). To ensure that venetoclax-treated CART induced antigen-specific killing, we repeated these cytotoxicity assays using CD19 null OCI-Ly8 tumor cells (OCI-Ly8.CD19KO) (Supplementary Fig. S2a). Venetoclax-treated CART failed to kill OCI-Ly8.CD19KO, indicating that CART-mediated killing was antigen-specific (Supplementary Fig. S2b). To test whether the enhanced cytotoxicity of venetoclax-treated CART was dependent upon having a certain co-stimulatory domain in the CAR construct, we compared killing by 4-1BB CD19CAR (used throughout this study) to an identical CD19CAR with a CD28 costimulatory domain. Additionally, to determine whether enhanced CART effector activity following venetoclax treatment is affected by different cytokines used during CART production, vehicle or venetoclax-treated CART were expanded in the presence of IL-2 (used throughout this study) or IL-7/IL-15. Venetoclax-treated CART induced similarly improved cytotoxicity in vitro regardless of which co-stimulatory molecule was incorporated into the CAR construct (Supplementary Fig. S2c), or the cytokine used during CART cell expansion (Supplementary Fig. S2d).
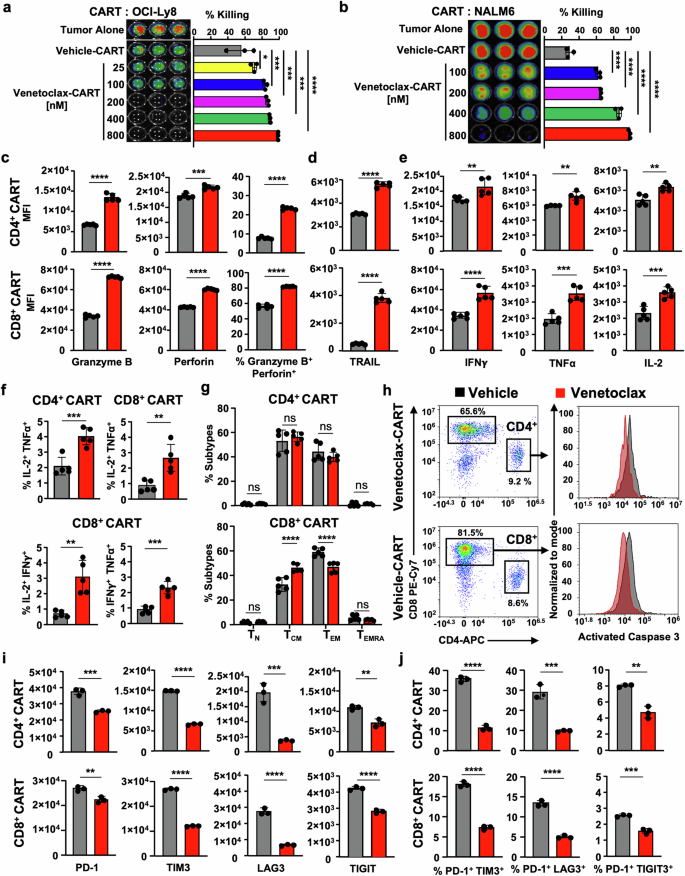
Venetoclax increases CART cytotoxicity in vitro, reflected in increased cytokine production, greater cell death resistance, and decreased exhaustion. Cytotoxicity of CART treated with vehicle or increasing doses of venetoclax against (a) CD19+ human DLBCL OCI-Ly8 and (b) CD19+ human acute lymphoblastic leukemia NALM6 cells using bioluminescent imaging quantification of remaining tumor cells after 48 h of co-culture at an E:T ratio of 1:5. Quantified results in adjacent bar graphs show that venetoclax dose-dependently enhances the anti-tumor efficacy of CART. c Venetoclax amplifies the killing function of CART via increased single and dual expression of granzyme B and perforin following stimulation with tumor target cells. d–f Venetoclax-treated CART upregulate expression of TNF-related apoptosis-inducing ligand (TRAIL) and have increased individual and co-expression of intracellular effector cytokines IFNγ, TNFα, and IL-2 following tumor stimulation. g Memory profiling shows no significant differences in CD4+ CART cell phenotype and indicates accumulation of TCM in venetoclax-treated CD8+ CART while more TEM are detected in vehicle-treated CD8+ CART. h Venetoclax-treated CART demonstrates longer persistence in vitro, marked by a reduced tendency to undergo apoptosis following encounter with OCI-Ly8 tumor cells. i, j Venetoclax-treated CART exhibit a less exhausted CART cell phenotype after two days of incubation with tumor cells. Data represent mean ± SD. Statistics for (a, b) one-way ANOVA with post-hoc Tukey’s tests were performed, unpaired Student’s t-test for (c–f, i–j), and two-way ANOVA with SIDAK’s multiple comparison tests for (g). a–j Results shown are from one representative experiment of at least three independent experiments with similar results using CART from three healthy donors. g Data are summarized from CART prepared from two healthy donors with two-three technical replicates in bar graphs and shown as mean ± SD. *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001, ns non-significant
Amplified T cell effector functions mediate venetoclax-treated CART enhanced cytotoxicity
To better understand the cytotoxic mechanisms responsible for venetoclax-mediated CART changes, we measured T cell effector cytokines, apoptosis, and exhaustion markers in venetoclax-treated CART following tumor antigen stimulation. Following co-culture with OCI-Ly8 cells, venetoclax-treated CART had greater expression of granzyme B, perforin, TRAIL, IFN-γ, TNF-α, and IL-2, all essential for CAR T-cell mediated cytotoxicity (Fig. 2c–f and Supplementary Fig. S3a). Cell death receptor/ligand pairs, including TRAIL/TRAIL-R1/R2, are known to play roles in CART cell effector killing through direct apoptosis induction of tumor cells.34,35,36 To test if TRAIL also played a significant role in venetoclax-treated CART-mediated killing, we performed neutralization experiments and found that antibody-mediated inhibition of TRAIL dose-dependently decreased venetoclax- and vehicle-treated CART efficacy (Supplementary Fig. S3b). We next wondered if venetoclax-related killing was reflected in an increased percentage of effector CART cells following tumor cell recognition. While the starting CART product from vehicle- and venetoclax-treated cells showed no differences in memory status (Fig. 1h), following tumor stimulation, there was a significant increase in the percentage of CD8+ central memory (TCM) and decreased percentage of CD8+ effector memory (TEM) cells within venetoclax-treated CART compared to the vehicle-treated product (Fig. 2g). Conversely, venetoclax-treated CD4+CART cells had no significant memory changes compared to vehicle-treated cells (Fig. 2g). Given these results, we next questioned if the increased anti-apoptotic protein expression measured in venetoclax-treated CART (Fig. 1c–e) may have led to less activation induced cell death (AICD), which can be associated with poor CART survival. Indeed, venetoclax-treated CART were more resistant to cell death following tumor target engagement compared to vehicle-treated cells, as reflected in less activated caspase 3 in these cells (Fig. 2h). The majority of these cells were CD8+ compared to CD4+ in both venetoclax- and vehicle-treated CART. However, venetoclax treatment led to a higher percentage of CD8+ cells compared to vehicle-treatment (81.5% vs 65.6%). In addition, compared to vehicle-treated cells, venetoclax-treated CART had significantly lower single and combined protein expression of canonical markers of T cell exhaustion, including PD-1, TIM3, LAG3, and TIGIT following co-culture with tumor cells (Fig. 2i, j). We wondered next if these results suggest that venetoclax-treated CART cells would kill target cells more efficiently and have prolonged serial killing ability. To test this, CART cells were incubated with OCI-Ly8 tumor cells at an effector:target (E:T) ratio of 1:1 and were serially challenged with tumor cells at the same E:T ratio until all tumor cells were killed. Venetoclax-treated CART cells were significantly better at killing tumor cells over time than vehicle-treated CART, while having similar proliferation in culture compared to vehicle-treated CART cells (Supplementary Fig. S3d). Thus, expansion of CART in the presence of therapeutic BCL-2 blockade improves their effector function, renders them more cell death resistant, and results in lower markers of exhaustion following interaction with target tumor cells.
Venetoclax-enhanced antitumor activity of CART is BCL-2 dependent
To evaluate whether the effects following venetoclax treatment in CART cells were due to on-target drugging of BCL-2, we designed CART vectors that overexpressed genes encoding either wild-type BCL-2 (BCL-2(WT)) or one of three BCL-2 constructs with mutations within the BH3 binding pocket known to incur various levels of venetoclax resistance.37,38,39 Of these mutations, BCL-2(F104L) has the least resistance to venetoclax binding compared to BCL-2(F104C) and BCL-2(G101V) mutations.39 We hypothesized that if our observations were specific to BCL-2 drugging, then attenuation of these effects would correlate with venetoclax resistance. We focused first on examining CART constructs with the most resistant forms of BCL-2 and compared them to CART overexpressing BCL-2(WT). There were no significant differences in the final product cell counts between vehicle- and venetoclax-treated CART, although CART with BCL-2(WT) showed a trend towards greater proliferation (Fig. 3a). BCL-2 protein levels were increased in all CART cells regardless of the BCL-2 isoform overexpressed and was uniformly increased following venetoclax treatment, as measured previously (Fig. 3b, c). Notably, however, the fold increase in other anti-apoptotic proteins was greatest in venetoclax-treated CART BCL-2(WT) compared to CART cells containing mutant isoforms of BCL-2. These data suggest that binding of venetoclax to the BCL-2 BH3 pocket is critical for the observed increases in BCL-XL and MCL-1. Next, to ensure equivalent comparisons of therapeutic efficacy, we measured no differences in CAR expression between the CART populations (Fig. 3d). In further support of venetoclax binding to BCL-2 as the on-target mechanism responsible for increased CART-mediated activity, tumor cell killing was highest by venetoclax-treated CART BCL-2(WT) and lowest by CART BCL-2(G101V) (Fig. 3e). Thereby, while venetoclax was still able to target endogenous wild-type BCL-2 within all CART constructs, killing was significantly attenuated in mutant BCL-2-containing CART cells. Furthermore, CART cells co-expressing mutant forms of BCL-2 regained an exhaustion phenotype similar to vehicle-treated cells following incubation with tumor cells (Fig. 3f). This finding was directly proportional to the relative affinities of BCL-2 for venetoclax. Collectively, these results confirm that the effects of venetoclax on CART are due to direct binding to the BCL-2 BH3-binding pocket. Moreover, these changes are due to alterations beyond apoptotic modulation and suggest alterations in other T cell signaling pathways.
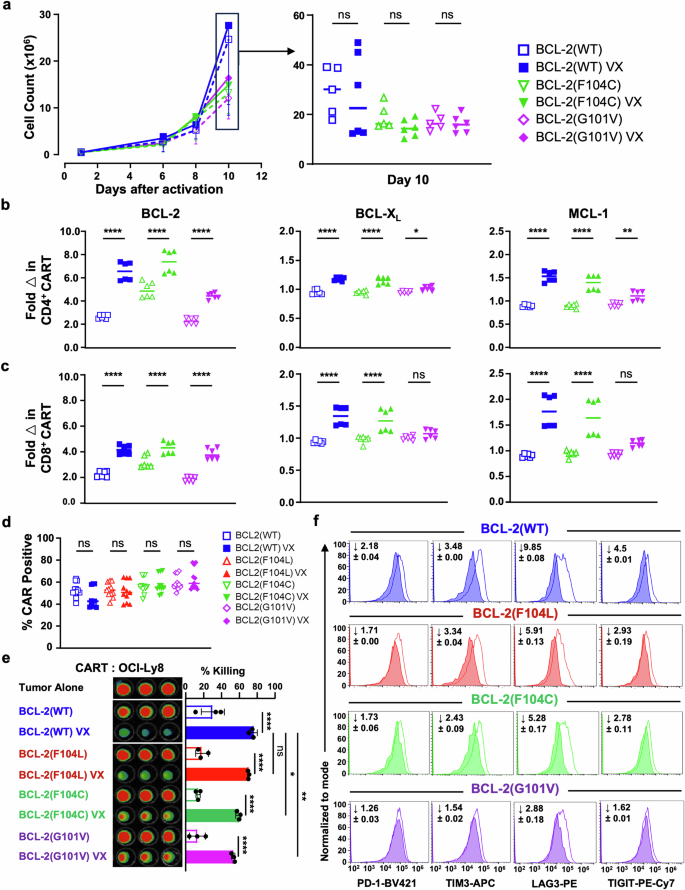
Venetoclax improves CART efficacy in a BCL-2-dependent manner. CART cells with various BCL-2 constructs treated with vehicle are indicated by an empty symbol, and venetoclax-treated CART (VX: 800 nM) are indicated by a filled symbol. a Proliferation curves of CART products at different time points. On right: Comparison of cell counts of each CD19CAR construct on day 10 of expansion demonstrates no significant changes in expansion between vehicle vs venetoclax-treated CART. b, c Anti-apoptotic BCL-2 family protein expression in different BCL-2 overexpressing CART constructs as measured flow cytometrically and normalized to the corresponding vehicle-treated CART by MFI fold change from products made from two healthy donors. BCL-2 expression was upregulated in all CART and further upregulated to different extents following treatment with venetoclax. CART with BCL-2 mutants shows decreased protein level changes of BCL-XL and MCL-1, correlating to their relative levels of venetoclax resistance. d CAR expression is comparable among T cells transduced with different CAR constructs and remained similar whether treated with vehicle or venetoclax. e CART expressing BCL-2 mutants shows attenuation of venetoclax-induced antitumor activity compared to venetoclax-treated CART expressing BCL-2(WT). f Venetoclax-treated CART with BCL-2 mutants show loss of venetoclax-related ability to resist exhaustion. A fold decrease in expression of PD-1, TIM3, LAG3, and TIGIT between venetoclax- and vehicle-treated CART is indicated in each histogram. a–c Data are from CART prepared from two healthy donors with three technical replicates, and MFI normalized to the corresponding vehicle-treated donor’s CART. d Data summarized from three independent experiments with three healthy donor samples and three technical replicates each. e, f Representative data from one experiment out of three independent experiments. a–d Data shown as mean ± SD. Two-way ANOVA with SIDAK’s multiple comparison tests was performed. *P < 0.05, **P < 0.01, ****P < 0.0001, ns non-significant
Venetoclax induces universal transcriptional changes in CART
Based on the above results, it appeared that the presence of BCL-2 and not alterations in the CD4+/CD8+ T cell ratio or memory phenotype was directly associated with the increased efficacy of venetoclax-treated CART cells. Therefore, we hypothesized that venetoclax induced gene expression reprogramming leading to enhanced T cell signaling and effector-like characteristics. To test this, we first performed bulk RNAseq on sorted CD4+ CAR+ and CD8+ CAR+ T cells prepared from PBMCs from three healthy donors following either vehicle or venetoclax treatment. Venetoclax led to distinct transcriptional profiles in both CD4+ and CD8+ CART cells as measured by principal component analysis (PCA) (Supplementary Fig. S4a) and resulted in alterations in genes representing similar signaling pathways within each cell type as determined by HALLMARK analysis (Supplementary Fig. S4b). We next sought to identify whether these gene expression changes were specific to certain CART cell subpopulations. In this context, we performed single-cell analysis (scRNAseq) on vehicle- or venetoclax-treated CART prepared from an additional three healthy donors to measure effects of venetoclax on CD4+ and CD8+ CART subsets (Fig. 4a, d). Venetoclax treatment did not significantly alter the cluster designation in either the CD4+ or CD8+ CART products (Fig. 4b, e). However, while there were no significant differences in the relative percentage of individual clusters between venetoclax- and vehicle-treated CART, there was a trend for venetoclax-treated cells to have signatures reflective of increased “proliferative” (cluster 1; e.g., MKI67) and less “naïve” (cluster 3; e.g., CCR7/TCF7/IL7R) states within CD8+ CART cells (Fig. 4b, e). Notably, BCL2 was significantly increased across all venetoclax-treated CD4+ and CD8+ CART cell clusters relative to vehicle-treated cells (Fig. 4c, f). scRNAseq identified 514 and 1078 differentially expressed genes (DEGs) in CD8+ and CD4+ T cells, respectively (Fig. 4g). Using gene set enrichment analysis (GSEA) against HALLMARK pathways, we found that venetoclax-treatment promoted the enrichment of similar pathways between CD4+ and CD8+ CART, suggesting a shared mechanism of action (Fig. 4h). Many of the significantly altered transcription programs were again associated with T cell activation, effector function, cell cycle, survival and metabolism highlighted by positive enrichment of genes associated with IL-2/STAT5, PI3K/AKT/MTOR, MTORC1, MYC, and TNFα/NFκB signaling (Fig. 4h). Significant enrichment of these pathways was consistent across CD4+ and CD8+ CART cell clusters (Fig. 4i). GSEA also demonstrated upregulation of transcriptomic signatures associated with IFN-α, IFN-γ and TNF-α signaling (Supplementary Fig. S4c). To correlate alterations in BCL-2 family protein levels (Fig. 1c–e) to transcriptional changes, we found several BCL-2 family transcript changes, including upregulation of BCL2, BCL2L1, BAX, and PMAIP1 (NOXA) and downregulation of MCL1 and BCL2L11 (BIM) in venetoclax-treated CART cells (Supplementary Fig. S4d). The upregulation of BCL2 and downregulation of BCL2L11 correspond with our previous work showing similar results in murine T cells treated with venetoclax.24,25 Low gene expression in the setting of MCL-1 protein increase (Fig. 1c–e) is a well described phenomena secondary to extension of its protein half-life following binding within its BH3 pocket, either following treatment with MCL-1-specific BH3 mimetics or through dissociation of BCL-2 family proteins from other anti-apoptotic family members and subsequent binding to MCL-1.40,41
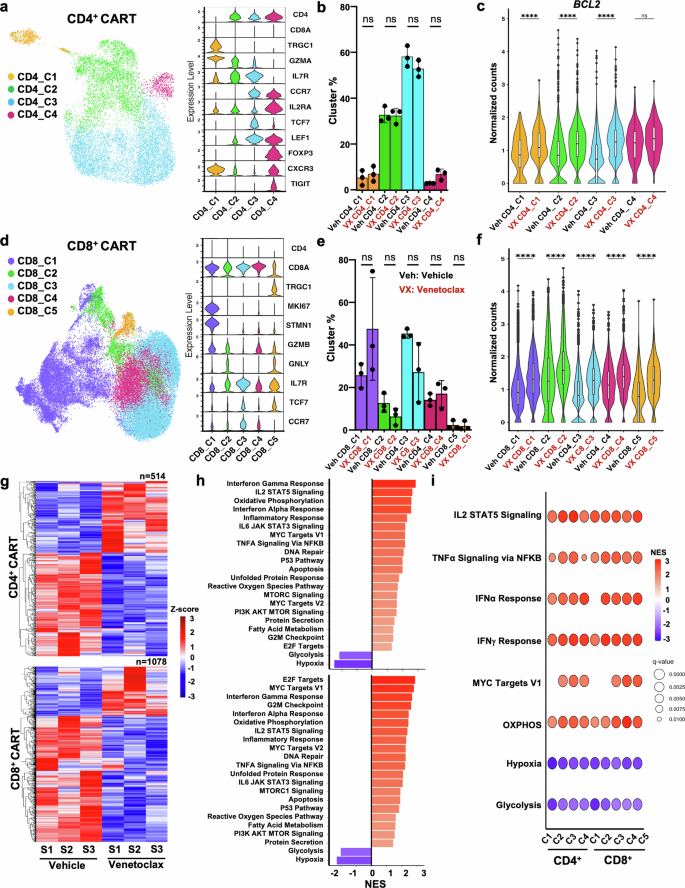
Gene expression analysis of venetoclax-treated CART reveals transcriptional T cell reprogramming. Vehicle or venetoclax-treated CART were generated as in Fig.1a, sorted for live cells, and analyzed by scRNAseq on day 13 after expansion. The experiment was performed using three biological replicates. a, d Uniform manifold approximation and projection (UMAP) embedding of clusters in CD4+ and CD8+ CART by scRNAseq. Key genes used for cluster designation are indicated. b, e Venetoclax-treated CART show similar cluster proportions compared to vehicle-treated cells. The proportion of each cluster within its respective treatment group are indicated by dots representing individual donors. c, f BCL2 transcript level compared between each cluster of CD4+ and CD8+ CART treated with vehicle or venetoclax shows that venetoclax upregulated BCL2 globally in all cell clusters. g Venetoclax-treatment led to a distinctive alteration in CART transcriptional profiles, as indicated by a heatmap of pseudo-bulk Z-score-scaled expression of DEGs with BH-adjusted P value < 0.05, avg_log2FC > 0.58. h Highly upregulated and downregulated pathways in venetoclax-treated CART compared to vehicle-treated CART. i HALLMARK pathway analysis across CART cell subpopulation clusters demonstrates global alterations. b, c, e, f Data shown as mean ± SD. Two-way ANOVA with SIDAK’s multiple comparison tests was performed. ****P < 0.0001, ns non-significant. Veh: Vehicle (black) and VX: venetoclax (red) in parts b, c, e, f
Given the common enriched pathways shared between scRNAseq and bulk RNAseq signatures of CD4+ and CD8+ CART populations, we sought to identify whether select genes were core contributors to these enrichments. To do this, we identified genes that were most frequently present in the leading edge of each enrichment across both sets of data among all six donors. We found that most genes were shared across datasets, suggesting that core transcriptomic mechanisms existed across conserved pathways following venetoclax treatment (Supplementary Fig. S5). Altogether, these data indicate that the enhanced effect observed with venetoclax in CART results from broad-spectrum transcriptional reprogramming of T cells that modulate T cell signaling, functionality, and metabolic fitness of the CART product.
The enhanced antitumor effect of venetoclax-treated CART is mediated in part by PI3K/AKT and JAK/STAT signaling
Recent data suggest that PI3K/AKT and JAK/STAT5 signaling are key determinants of CART fitness, as defined by increased proliferation, effector function, memory, and survival.30,42,43,44 We previously reported that homeostatically expanding naïve murine CD4+ and CD8+ T cells acquire effector-like gene signatures following in vivo administration of venetoclax, marked by upregulation of PI3K/AKT and JAK/STAT signaling pathways.24 Aligned with this data, GSEA of scRNAseq of venetoclax-treated CART revealed a significant enrichment of genes associated with PI3K/AKT/MTOR and IL-2/STAT5 signaling (Fig. 5a, b). To confirm increased signaling within these pathways, CART cells were evaluated for the active phosphorylated forms of STAT5 and AKT. Western blot analysis showed that total CART expanded in the presence of venetoclax had increased pSTAT5 and pAKT when compared to vehicle-treated CART (Supplementary Fig. S6b, part I). This was also confirmed flow cytometrically in individual CD4+ and CD8+ CART cells (Fig. 5a, b). We then used pharmacologic inhibitors of STAT5 and AKT to evaluate if these pathways were responsible for the increased efficacy of venetoclax-treated CART products. Here, CART were expanded as detailed in Fig. 1a and then treated with either AC-4-130 (STAT5 inhibitor) or AKT inhibitor VIII (AKT 1/2 inhibitor) for 24 h prior to incubation with OCI-Ly8 cells.45,46 STAT5 inhibition significantly reduced venetoclax-treated CART-mediated killing to a greater extent than vehicle-treated CART (Fig. 5c and Supplementary Fig. S6a). We confirmed that STAT5 inhibition decreased pSTAT5 and pAKT in these cells (Supplementary Fig. S6b, part II). AKT inhibition also reduced the efficacy of venetoclax-treated CART (Fig. 5d). However, in contrast to STAT5 inhibition, and aligned with previously published reports, AKT inhibition resulted in increased efficacy of vehicle-treated CART (Fig. 5d).47,48 Assessment of these CART products indicated a greater decrease in pAKT in venetoclax-treated CART compared to vehicle-treated CART (Supplementary Fig. S6b, part III). Neither treatment resulted in changes in BCL-2 protein levels. Collectively, these results indicate a reliance on both STAT5 and AKT pathways for venetoclax-mediated effects on CART cell killing.
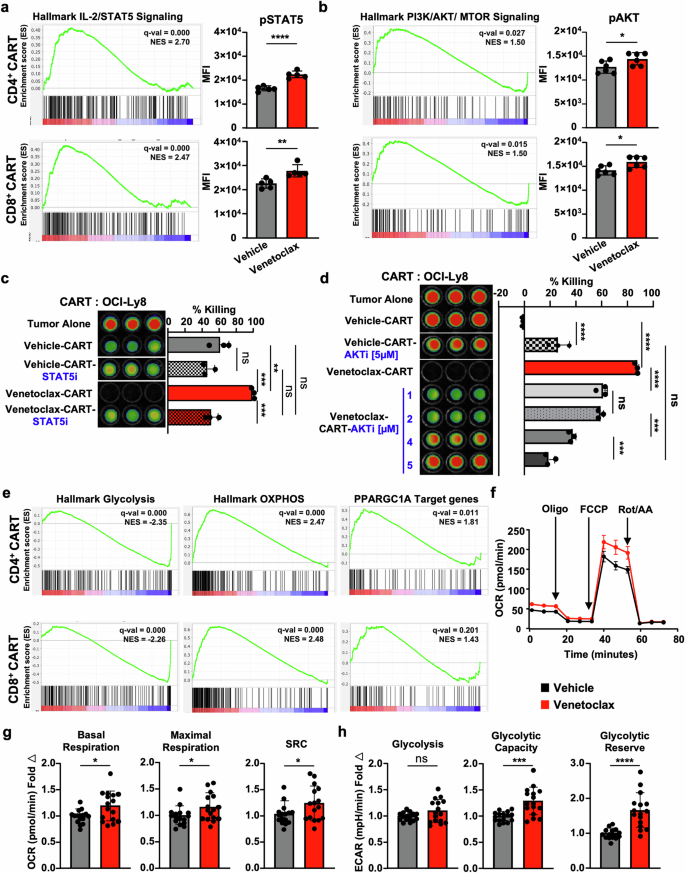
Venetoclax alters CART cell signaling and improves metabolic fitness. a, b Venetoclax-treatment results in transcriptional changes associated with IL-2/STAT5 and PI3K/AKT/MTOR signaling as indicated by GSEA of CD4+ and CD8+ CART scRNAseq. These changes are reflected in increased intracellular protein expression of (a) phospho-STAT5 (Tyr694) and b phospho-AKT (Ser473) in venetoclax-treated compared to vehicle-treated CART. Therapeutic inhibition of (c) STAT5 and (d) AKT reverses the increased killing effect following venetoclax treatment as measured using in vitro tumor killing assays against OCI-Ly8 DLBCL cells. Effector to tumor (E:T) ratio of 1:5 and incubation for 48 h prior to imaging. e Venetoclax-treatment results in transcriptional changes associated with decreased glycolysis and increased OXPHOS and PPARGC1A-related signaling as indicated by GSEA of CD4+ and CD8+ CART scRNAseq. f Venetoclax-treated CART cells have higher OXPHOS as measured by increased oxygen consumption rate (OCR). Venetoclax-treated CART also have increased (g) basal respiration, maximal respiration, and spare respiratory capacity (SRC) while still maintaining (h) higher glycolytic capacity and reserve. Seahorse data represent analysis of n = 8 technical replicates from two healthy donors of two independent experiments in mean ± SD (g, h) and mean ± SEM (f). Unpaired Student’s t-test was performed for (a, b, g, h), and one-way ANOVA with post-hoc Tukey’s tests was performed for (c, d)
Venetoclax promotes CART metabolic fitness
HALLMARK analyses of venetoclax-treated CART also showed positive enrichment of pathways associated with fatty acid metabolism, OXPHOS, and ROS and negative enrichment of signatures associated with glycolysis and hypoxia in both CD4+ and CD8+ CART, as well as distinct enrichment signatures in PPARGC1A (Figs. 4h and 5e). PPARGC1A encodes for PGC1α, a key metabolic regulator that promotes mitochondrial biogenesis, OXPHOS, and fatty acid β-oxidation, and results in enhanced T cell fitness, memory formation, and T cell-mediated antitumor potency.49 Therefore, we sought to determine if there was a connection between these transcriptional alterations and actual metabolic profiles of venetoclax-treated CART. To test this, the oxygen consumption rate (OCR) of vehicle- and venetoclax-treated CART following expansion was measured using Seahorse Mito Stress analyses.50 Venetoclax significantly increased basal OCR, maximal respiration, and spare respiratory capacity (SRC) in CART (Fig. 5f, g).
OCR is considered a primary source of ROS in T cells.51,52 Related to this, venetoclax has been found to enhance the antileukemic effector activity of patient-derived T cells through increased ROS due to impaired respiratory chain supercomplex formation. In that setting, increased T cell-mediated killing was inhibited by the antioxidant N-acetyl-L-cysteine (LNAC).22 Thus, we wondered if the killing capacity of venetoclax-treated CART depended on upregulated ROS production that could be linked to enhanced OXPHOS levels. To test this, we incubated venetoclax-treated CART with escalating doses of LNAC and found no effect on their tumor killing capacity (Supplementary Fig. S6c). Thereby, ROS induction does not appear to be involved in the effect of venetoclax on CART in our system.
We next measured the extracellular acidification rate (ECAR) of venetoclax-treated CART to determine if their glycolytic status differed from vehicle-treated CART. While glycolysis was not affected at baseline, venetoclax-treated CART had significantly elevated glycolytic capacity and glycolytic reserve compared to vehicle-treated cells (Fig. 5h). Additionally, transcriptional data revealed that venetoclax treatment upregulated key genes involved in OXPHOS and fatty acid metabolism (e.g., CPT1A and FABP5) and downregulated genes associated with glycolysis (e.g., PDK1, PGK1, SLC16A3, and VEGFA), a pattern well known to increase CART persistence (Supplementary Fig. S7).53
Balancing of STAT5 and AKT signaling has been associated with differential metabolic effects on CART and effector T cells.47,48,54,55,56 To determine if STAT5 or AKT pathways were responsible for the metabolic changes in venetoclax-treated CART, Seahorse analyses were performed on CART in the setting of STAT5 or AKT inhibition (Supplementary Fig. S6d). In contrast to similar globally decreased metabolic effects following AKT inhibition in vehicle- and venetoclax-treated CART, as previously reported,47 STAT5 inhibition led to significant decreases in OXPHOS and glycolysis-related measurements only in venetoclax-treated CART, indicating that STAT5 signaling is primarily responsible for the observed venetoclax-mediated metabolic effects.
Venetoclax-treated CART prepared from healthy donors has increased therapeutic activity against leukemia and lymphoma in vivo
Given the measured improvement of venetoclax-treated CART in vitro, we next sought to investigate whether these effects would translate in vivo. To measure this, two CD19+ xenograft tumor models were used, disseminated NALM6 leukemia and subcutaneously injected OCI-Ly8 DLBCL (Fig. 6a, d). In both cases, mice that received venetoclax-treated CART had delayed tumor growth (Fig. 6b, e) that translated into significantly improved survival (Fig. 6c, e). Vehicle- and venetoclax-treated CART could be detected in animals prior to reaching advanced stages of disease. There were greater ratios of TEM CD8+ CART following tumor challenge in those animals that received venetoclax-treated CART compared to more terminally differentiated (TEMRA) CART in animals that received vehicle-treated CART (Supplementary Fig. S8)
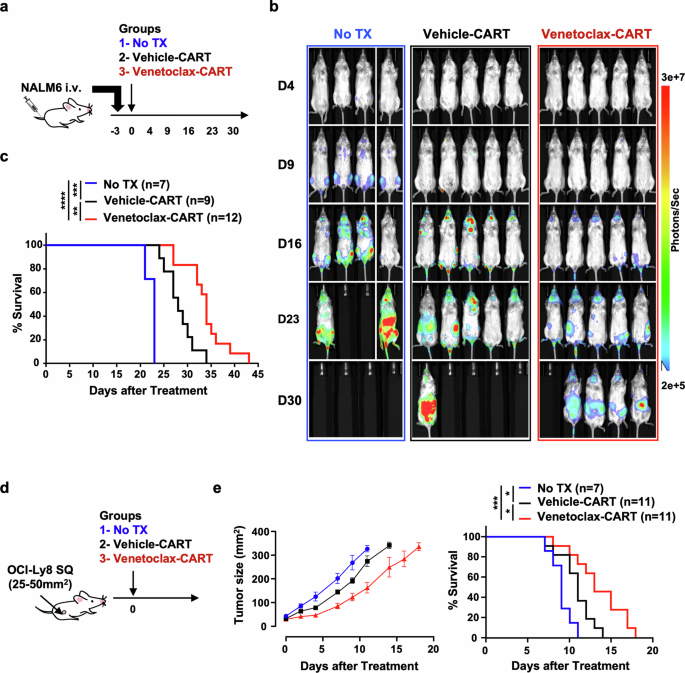
Venetoclax augments the in vivo efficacy of CART prepared from healthy donors. a Treatment schema for the NALM6 leukemia xenograft model. Three days following NALM6 injection, mice were randomly divided into groups that either received no treatment (No TX), vehicle-treated CART, or venetoclax-treated CART. b, c Animals that received venetoclax-treated CART had improved tumor control, resulting in significantly prolonged survival. d Treatment schema for the OCI-Ly8 DLBCL xenograft model. Mice with established subcutaneous (SQ) tumors (25–50 mm2) received no treatment (No TX), vehicle-treated CART, or venetoclax-treated CART. e Animals that received venetoclax-treated CART had improved tumor control and significantly prolonged survival. These results are pooled from three independent experiments. Data shown as mean ± SD. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001
Venetoclax amplifies the therapeutic potency of CART prepared from patient-derived T cells
We next tested if venetoclax could enhance the efficacy of CART prepared from chemotherapeutically pretreated patient-derived T cells that were isolated at the time of apheresis for commercial CD19+ CART products (Supplementary Table S1). Starting material from each sample was flow cytometrically characterized for its CD4:CD8 ratio, Treg content, memory phenotype, exhaustion markers, and anti-apoptotic protein expression. CD4:CD8 ratios between samples were markedly different, while the percentages of FOXP3+ CD4+ Tregs were generally similar between samples (Fig. 7a, b and Supplementary Fig. S9a, b). The relative percentages of naive (CCR7+, CD45RA+), central memory (CCR7+, CD45RA−), effector memory (CCR7−, CD45RA−), and effector memory RA+ (TEMRA, CCR7−, CD45RA+) T cells across samples showed that patient samples universally had fewer naïve T cells compared to those from healthy volunteers (Fig. 7c and Supplementary Fig. S9c). Furthermore, patient-derived CD4+ and CD8+ T cells had increased expression of T cell exhaustion markers, particularly LAG3 and PD-1, compared to those from healthy donors, and had less memory characteristics reflected by decreased levels of CD62L, CD127, and TCF1 (Fig. 7d, e and Supplementary Fig. S9d). There were also heterogeneous baseline protein levels of BCL-2, BCL-XL, and MCL-1 between the samples (Fig. 7f and Supplementary Fig. S9e). BCL-2 protein levels were particularly different across samples, which may be pertinent given the recent finding that BCL-2 expression in T cells from patient apheresis products positively correlated with CART response.28 While there were differences in overall CAR transduction efficiency between samples, there were no significant differences in CAR expression between vehicle- and venetoclax-treated samples within individual subjects (Supplementary Figs. S9f and S10a). There were, however, marked differences between the final CART cell CD4:CD8 ratios between donors, which were somewhat reflective of their starting T cell ratios (Fig. 7a and Supplementary Fig. S10b). Not surprisingly, there was a greater percentage of CAR+ FOXP3+ Tregs in CART products manufactured from T cells collected from patients compared to healthy donors. Venetoclax treatment did not affect these percentages (Supplementary Fig. S10c). Despite differences in starting material and post-production CD4+:CD8+ CART cell ratios, venetoclax treatment significantly enhanced the in vitro and in vivo CART efficacy of all patient-derived CART products (Fig. 7g, h and Supplementary Fig. S9g). These results indicate that venetoclax treatment during ex vivo expansion can reprogram CART cells and increase their efficacy irrespective of the starting T cell material. These results warrant further investigation into the use of venetoclax to reprogram patient-derived T cells for use in CART-related and other adoptive T cell treatments.
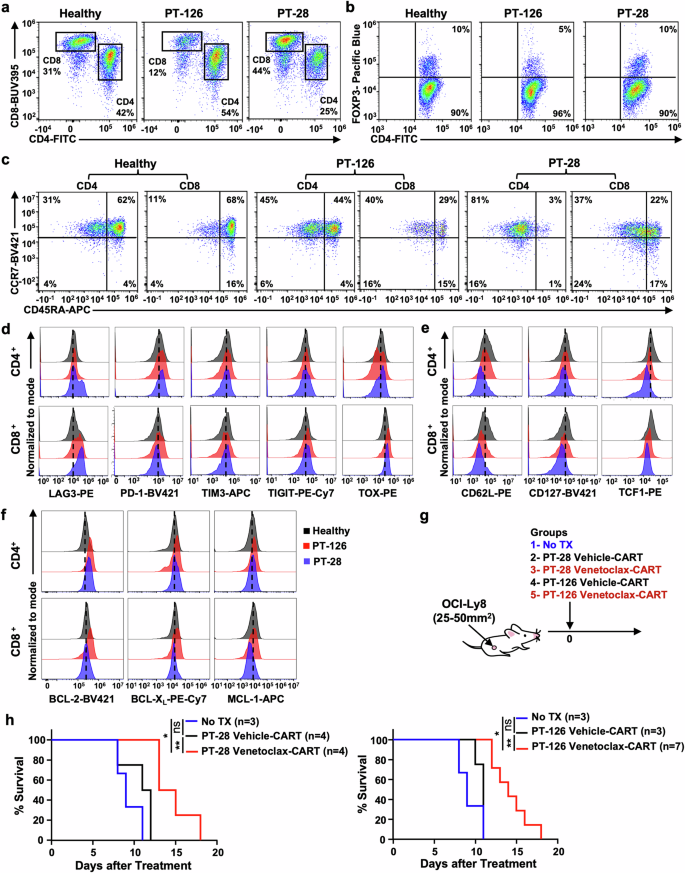
Venetoclax enhances the potency of CART prepared from patient samples regardless of their starting T cell composition, memory phenotype, or exhaustion profiles. Patient-derived PBMCs (PT-28 and PT-126) collected at the time of commercial CART cell apheresis were used to isolate T cells and prepare vehicle or venetoclax-treated CART. PBMCs from healthy volunteers were used as comparators. Starting material was evaluated for (a) CD4:CD8 ratio, b CD4+ FOXP3+ Treg content, c CD4+ and CD8+ T cell subpopulation content as determined by CCR7 and CD45RA, d exhaustion phenotype based on surface expression of LAG3, PD-1, TIM3, TIGIT and intracellular marker TOX, e T cells memory status based on the surface expression of CD62L and CD127 and intracellular expression of TCF-1, and f protein expression levels of BCL-2, BCL-XL, and MCL-1. g Mice with established subcutaneous OCI-Ly8 tumors (25–50 mm2) received no treatment (No TX), vehicle-treated CART, or venetoclax-treated CART as indicated. h Treatment with venetoclax-treated CART prepared from patient samples (PT-28 or PT-126) resulted in prolonged survival compared to vehicle-treated CART. The number of mice in each group is given. Data shown as mean ± SD. *P < 0.05, **P < 0.01, ns non-significant















Leave a Reply