de Perrot, M., Liu, M., Waddell, T. K. & Keshavjee, S. Ischemia-reperfusion-induced lung injury. Am. J. Respir. Crit. Care Med. 167, 490–511 (2003).
Dugbartey, G. J. Therapeutic benefits of nitric oxide in lung transplantation. Biomed. Pharmacother. Biomed. Pharmacother. 167, 115549 (2023).
Xu, L. F. et al. Extracorporeal membrane oxygenation during double-lung transplantation: single center experience. Chin. Med. J. 123, 269–273 (2010).
Chacon-Alberty, L. et al. Primary graft dysfunction in lung transplantation: a review of mechanisms and future applications. Transplantation 107, 1687–1697 (2023).
Zhao, M. et al. Alveolar macrophage activation is a key initiation signal for acute lung ischemia-reperfusion injury. Am. J. Physiol. Lung Cell Mol. Physiol. 291, L1018–L1026 (2006).
Locati, M., Curtale, G. & Mantovani, A. Diversity, mechanisms, and significance of macrophage plasticity. Annu. Rev. Pathol. 15, 123–147 (2020).
Sica, A. & Mantovani, A. Macrophage plasticity and polarization: in vivo veritas. J. Clin. Investig. 122, 787–795 (2012).
Mahon, O. R. et al. Nano-particle mediated M2 macrophage polarization enhances bone formation and MSC osteogenesis in an IL-10 dependent manner. Biomaterials 239, 119833 (2020).
Wang, Y. H. et al. Gut microbiota-derived succinate aggravates acute lung injury after intestinal ischaemia/reperfusion in mice. Eur. Respir. J. 61, 2200840 (2023).
Liu, H. et al. Efficacy of pulmonary transplantation of engineered macrophages secreting IL-4 on acute lung injury in C57BL/6J mice. Cell Death Dis. 10, 664 (2019).
Yang, J. et al. Extracellular vesicles derived from M2-like macrophages alleviate acute lung injury in a miR-709-mediated manner. J. Extracell. Vesicles 13, e12437 (2024).
Matthay, M. A. et al. Treatment with allogeneic mesenchymal stromal cells for moderate to severe acute respiratory distress syndrome (START study): a randomised phase 2a safety trial. Lancet Respir. Med. 7, 154–162 (2019).
Wick, K. D., Leligdowicz, A., Zhuo, H., Ware, L. B. & Matthay, M. A. Mesenchymal stromal cells reduce evidence of lung injury in patients with ARDS. JCI Insight 6, e148983 (2021).
Brennan, P. J., Brigl, M. & Brenner, M. B. Invariant natural killer T cells: an innate activation scheme linked to diverse effector functions. Nat. Rev. Immunol. 13, 101–117 (2013).
Crosby, C. M. & Kronenberg, M. Tissue-specific functions of invariant natural killer T cells. Nat. Rev. Immunol. 18, 559–574 (2018).
Dasgupta, S. & Kumar, V. Type II NKT cells: a distinct CD1d-restricted immune regulatory NKT cell subset. Immunogenetics 68, 665–676 (2016).
Lu, H. et al. Potent NKT cell ligands overcome SARS-CoV-2 immune evasion to mitigate viral pathogenesis in mouse models. PLoS Pathog. 19, e1011240 (2023).
Bayatipoor, H. et al. Role of NKT cells in cancer immunotherapy-from bench to bed. Med. Oncol. 40, 29 (2022).
Halder, R. C., Aguilera, C., Maricic, I. & Kumar, V. Type II NKT cell-mediated anergy induction in type I NKT cells prevents inflammatory liver disease. J. Clin. Investig. 117, 2302–2312 (2007).
Arrenberg, P., Maricic, I. & Kumar, V. Sulfatide-mediated activation of type II natural killer T cells prevents hepatic ischemic reperfusion injury in mice. Gastroenterology 140, 646–655 (2011).
Yang, S. H. et al. Sulfatide-reactive natural killer T cells abrogate ischemia-reperfusion injury. J. Am. Soc. Nephrol. 22, 1305–1314 (2011).
Pan, H. et al. Sulfatide-activated type II NKT cells suppress immunogenic maturation of lung dendritic cells in murine models of asthma. Am. J. Physiol. Lung Cell Mol. Physiol. 317, L578–l590 (2019).
Ni, H. et al. Role of sulfatide-reactive vNKT cells in promoting lung Treg cells via dendritic cell modulation in asthma models. Eur. J. Pharmacol. 970, 176461 (2024).
Maricic, I., Halder, R., Bischof, F. & Kumar, V. Dendritic cells and anergic type I NKT cells play a crucial role in sulfatide-mediated immune regulation in experimental autoimmune encephalomyelitis. J. Immunol. 193, 1035–1046 (2014).
Lin, Q. et al. Anti-CD1d treatment suppresses immunogenic maturation of lung dendritic cells dependent on lung invariant natural killer T cells in asthmatic mice. Int. Immunopharmacol. 124, 110921 (2023).
Lombardo, L. J. et al. Discovery of N-(2-chloro-6-methyl- phenyl)-2-(6-(4-(2-hydroxyethyl)- piperazin-1-yl)-2-methylpyrimidin-4- ylamino)thiazole-5-carboxamide (BMS-354825), a dual Src/Abl kinase inhibitor with potent antitumor activity in preclinical assays. Journal. Med. Chem. 47, 6658–6661 (2004).
Matute-Bello, G. et al. An official American Thoracic Society workshop report: features and measurements of experimental acute lung injury in animals. Am. J. Respir. Cell Mol. Biol. 44, 725–738 (2011).
Sharma, A. K. et al. Natural killer T cell-derived IL-17 mediates lung ischemia-reperfusion injury. Am. J. Respir. Crit. Care Med. 183, 1539–1549 (2011).
Lee, Y. J. et al. Simvastatin treatment boosts benefits of apoptotic cell infusion in murine lung fibrosis. Cell Death Dis. 8, e2860 (2017).
Kechichian, T. B., Shea, J. & Del Poeta, M. Depletion of alveolar macrophages decreases the dissemination of a glucosylceramide-deficient mutant of Cryptococcus neoformans in immunodeficient mice. Infect. Immun. 75, 4792–4798 (2007).
Choi, J. P. et al. An important role of tumor necrosis factor receptor-2 on natural killer T cells on the development of dsRNA-enhanced Th2 cell response to inhaled allergens. Allergy 69, 186–198 (2014).
Wang, Y. et al. Overexpression of TIM-3 in macrophages aggravates pathogenesis of pulmonary fibrosis in mice. Am. J. Respir. Cell Mol. Biol. 61, 727–736 (2019).
Di, A. et al. The TWIK2 potassium efflux channel in macrophages mediates NLRP3 inflammasome-induced inflammation. Immunity 49, 56–65.e54 (2018).
Menson, K. E. et al. Therapeutic efficacy of IL-17A neutralization with corticosteroid treatment in a model of antigen-driven mixed-granulocytic asthma. Am. J. Physiol. Lung Cell Mol. Physiol. 319, L693–l709 (2020).
Li, Q. & Nie, H. Advances in lung ischemia/reperfusion injury: unraveling the role of innate immunity. Inflamm. Res. 73, 393–405 (2024).
Liang, L. et al. Inhibition of YAP1 activity ameliorates acute lung injury through promotion of M2 macrophage polarization. MedComm. 4, e293 (2023).
Zhang, G. et al. Sulfatide-activated type II NKT cells prevent allergic airway inflammation by inhibiting type I NKT cell function in a mouse model of asthma. Am. J. Physiol. Lung Cell Mol. Physiol. 301, L975–L984 (2011).
Hoteit, L. et al. Macrophage switching: polarization and mobilization after trauma. Shock 59, 232–238 (2023).
Acheampong, K. K. et al. Subcellular detection of SARS-CoV-2 RNA in human tissue reveals distinct localization in alveolar type 2 pneumocytes and alveolar macrophages. mBio 13, e0375121 (2021).
Valdez, H. A. et al. Human macrophage polarization shapes B. pertussis intracellular persistence. J. Leukoc. Biol. 112, 173–184 (2022).
Kane, H. et al. Longitudinal analysis of invariant natural killer T cell activation reveals a cMAF-associated transcriptional state of NKT10 cells. eLife 11, e76586 (2022).
Parekh, V. V. et al. Glycolipid antigen induces long-term natural killer T cell anergy in mice. J. Clin. Investig. 115, 2572–2583 (2005).
Bortoluzzi, S. et al. Brief homogeneous TCR signals instruct common iNKT progenitors whose effector diversification is characterized by subsequent cytokine signaling. Immunity 54, 2497–2513.e2499 (2021).
Redin, E. et al. SRC family kinase (SFK) inhibitor dasatinib improves the antitumor activity of anti-PD-1 in NSCLC models by inhibiting Treg cell conversion and proliferation. J. Immunother. Cancer 9, e001496 (2021).
Wang, L. et al. Manipulation of macrophage polarization by peptide-coated gold nanoparticles and its protective effects on acute lung injury. J. Nanobiotechnol. 18, 38 (2020).
Nakamura, T. et al. Depletion of alveolar macrophages by clodronate-liposomes aggravates ischemia-reperfusion injury of the lung. J. Heart Lung Transplant. 24, 38–45 (2005).
Prakash, A. et al. Alveolar macrophages and Toll-like receptor 4 mediate ventilated lung ischemia reperfusion injury in mice. Anesthesiology 117, 822–835 (2012).
Hsiao, H. M. et al. Spleen-derived classical monocytes mediate lung ischemia-reperfusion injury through IL-1β. J. Clin. Investig. 128, 2833–2847 (2018).
Liu, Y. et al. Noninvasive imaging of CCR2(+) cells in ischemia-reperfusion injury after lung transplantation. Am. J. Transplant. 16, 3016–3023 (2016).
Maus, U. A. et al. Monocytes are potent facilitators of alveolar neutrophil emigration during lung inflammation: role of the CCL2-CCR2 axis. J. Immunol. 170, 3273–3278 (2003).
Zhou, P. et al. Monocytes promote pyroptosis of endothelial cells during lung ischemia-reperfusion via IL-1R/NF-κB/NLRP3 signaling. Life Sci. 276, 119402 (2021).
Patel, O. et al. Recognition of CD1d-sulfatide mediated by a type II natural killer T cell antigen receptor. Nat. Immunol. 13, 857–863 (2012).
Krijgsman, D., Hokland, M. & Kuppen, P. J. K. The role of natural killer T cells in cancer-a phenotypical and functional approach. Front. Immunol. 9, 367 (2018).
Cope, A., Le Friec, G., Cardone, J. & Kemper, C. The Th1 life cycle: molecular control of IFN-γ to IL-10 switching. Trends Immunol. 32, 278–286 (2011).
Cardone, J. et al. Complement regulator CD46 temporally regulates cytokine production by conventional and unconventional T cells. Nat. Immunol. 11, 862–871 (2010).
Rogers, N. C. et al. Syk-dependent cytokine induction by Dectin-1 reveals a novel pattern recognition pathway for C type lectins. Immunity 22, 507–517 (2005).
Akira, S. & Takeda, K. Toll-like receptor signalling. Nat. Rev. Immunol. 4, 499–511 (2004).
Hu, X. et al. IFN-gamma suppresses IL-10 production and synergizes with TLR2 by regulating GSK3 and CREB/AP-1 proteins. Immunity 24, 563–574 (2006).
Sun, Z. et al. Exosomal circPOLQ promotes macrophage M2 polarization via activating IL-10/STAT3 axis in a colorectal cancer model. J. Immunother. Cancer 12, e008491 (2024).
Zhao, J., Weng, X., Bagchi, S. & Wang, C. R. Polyclonal type II natural killer T cells require PLZF and SAP for their development and contribute to CpG-mediated antitumor response. Proc. Natl. Acad. Sci. USA 111, 2674–2679 (2014).
Hwang, D. et al. YAP promotes global mRNA translation to fuel oncogenic growth despite starvation. Exp. Mol. Med. 56, 2202–2215 (2024).
Byles, V. et al. The TSC-mTOR pathway regulates macrophage polarization. Nat. Commun. 4, 2834 (2013).
Zhu, L. et al. TSC1 controls macrophage polarization to prevent inflammatory disease. Nat. Commun. 5, 4696 (2014).
Han, R. et al. RAD001 (everolimus) attenuates experimental autoimmune neuritis by inhibiting the mTOR pathway, elevating Akt activity and polarizing M2 macrophages. Exp. Neurol. 280, 106–114 (2016).
Ip, W. K. E., Hoshi, N., Shouval, D. S., Snapper, S. & Medzhitov, R. Anti-inflammatory effect of IL-10 mediated by metabolic reprogramming of macrophages. Science 356, 513–519 (2017).
Li, K., Zhang, Y., Diao, Y. & Fan, S. Paeonol regulates the DDIT4-mTOR signaling pathway in macrophages to promote diabetic wound healing. Int. Immunopharmacol. 151, 114347 (2025).
Yang, H. L. et al. A defective lysophosphatidic acid-autophagy axis increases miscarriage risk by restricting decidual macrophage residence. Autophagy 18, 2459–2480 (2022).
Shi, L. et al. ETV5-mediated transcriptional repression of DDIT4 blocks macrophage pro-inflammatory activation in diabetic atherosclerosis. Cardiovasc. Toxicol. 25, 379–394 (2025).
Britto, F. A., Dumas, K., Giorgetti-Peraldi, S., Ollendorff, V. & Favier, F. B. Is REDD1 a metabolic double agent? Lessons from physiology and pathology. Am. J. Physiol. Cell Physiol. 319, C807–c824 (2020).
Wang, X. et al. Exosomal lncRNA HMMR-AS1 mediates macrophage polarization through miR-147a/ARID3A axis under hypoxia and affects the progression of hepatocellular carcinoma. Environ. Toxicol. 37, 1357–1372 (2022).
Ellisen, L. W. Growth control under stress: mTOR regulation through the REDD1-TSC pathway. Cell Cycle 4, 1500–1502 (2005).
Bedel, R. et al. Lower TCR repertoire diversity in Traj18-deficient mice. Nat. Immunol. 13, 705–706 (2012).





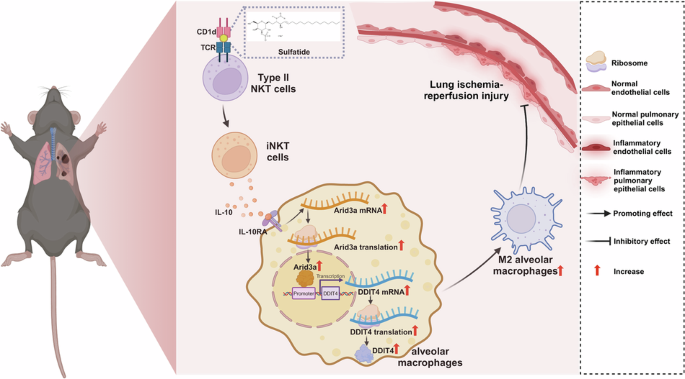









Leave a Reply