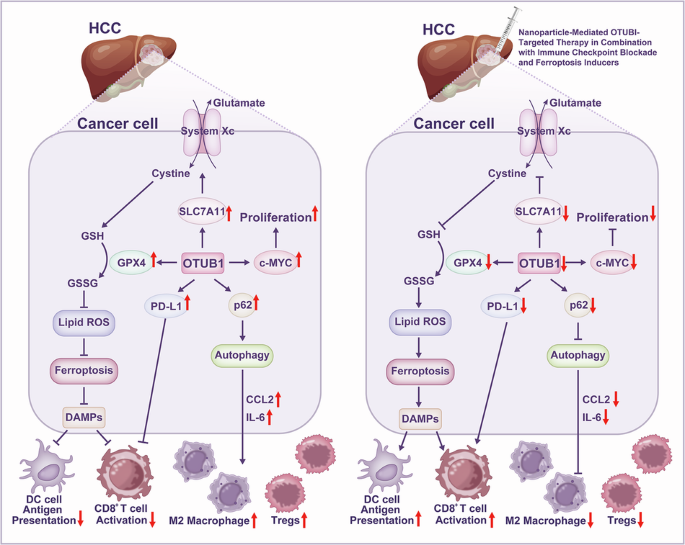
Hepatocellular carcinoma (HCC) treatment faces dual challenges: resistance to targeted therapy and low response rates to immunotherapy. These issues are rooted in the immunosuppressive tumor microenvironment (TME). Ferroptosis and autophagy, two critical cellular processes, play complex and paradoxical roles in HCC drug resistance and immunoregulation, and they interact closely. This review explores how the OTU deubiquitinase family, especially OTUB1, acts as a central hub coordinating the autophagy-ferroptosis balance. Additionally, other OTU family members, such as OTUD3, OTULIN, and OTUD6B, contribute to HCC progression by modulating similar pathways, highlighting the need for a broader therapeutic approach. Specifically, OTUD3 suppresses HIF-1α-driven angiogenesis, OTULIN inhibits NF-κB-mediated inflammation, and OTUD6B stabilizes pVHL to impede metastasis, collectively demonstrating their synergistic or antagonistic interactions with OTUB1 in reshaping the TME. This coordination drives HCC drug resistance and remodels the immune microenvironment. OTUB1 suppresses ferroptosis and maintains tumor cell survival by deubiquitinating and stabilizing key proteins like SLC7A11, GPX4, and p62. It also promotes immune escape by modulating PD-L1 stability and immune cell function. Consequently, therapeutic strategies targeting the OTU family—such as developing selective inhibitors for multiple members, using intelligent nanodelivery systems, and combining them with ferroptosis inducers or immune checkpoint inhibitors—show significant potential for reversing drug resistance and improving immunotherapy efficacy. Expanding these strategies to include other OTU members could enhance efficacy and reduce resistance. Addressing how the OTU family precisely modulates the intersection of autophagy and ferroptosis, and how it reshapes immune cell metabolism and function within the TME, is critical for developing novel combination therapies. This article provides a crucial theoretical foundation for developing novel combination strategies targeting metabolism-immune crosstalk.












Leave a Reply